The association between cannabis and appetite is well known both in the scientific world and pop culture.
As many of our longtime blog readers already know, this association ultimately led to the development of one of the most horrific weight loss drugs of the past century, known as Rimonabant. Researchers at the Sanofi-Aventis pharmaceutical company, noticing the role of THC (and more generally, activation of the CB1 cannabinoid receptor) in increasing appetite, guessed correctly that the reverse would also be true – that deactivating the CB1 receptor would decrease appetite and therefore allow weight loss. Of course, that was before scientists understood how essential the endocannabinoid system is to our healthy functioning; as numerous reports came flooding in of increased suicide rates and depression in individuals with no previous history of mental illness, the drug was quickly pulled from the market. Needless to say, plans for all similar CB1 blockers (CB1 antagonists) were nixed at competing pharmaceutical companies, and researchers have since been hesitant to touch cannabinoids as weight loss drugs, for good reason.
However, as we’ve expressed in previous articles, the endocannabinoid system is not a simple system compromised of only two light switches (in this case, two receptors that can merely be turned off or on). In fact, the endocannabinoid system can be activated through non-standard receptor binding sites, and endocannabinoids themselves can also activate other chemical pathways throughout the body. Unfortunately, due to a lack of research, many of the specific effects of manipulating the endocannabinoid system remain unknown. Regardless, the endocannabinoid system is well-situated to control weight loss via at least some mechanisms of action. This month, a review in the scientific journal Trends in Endocrinology and Metabolism outlines the multiple ways the system interacts with weight loss.
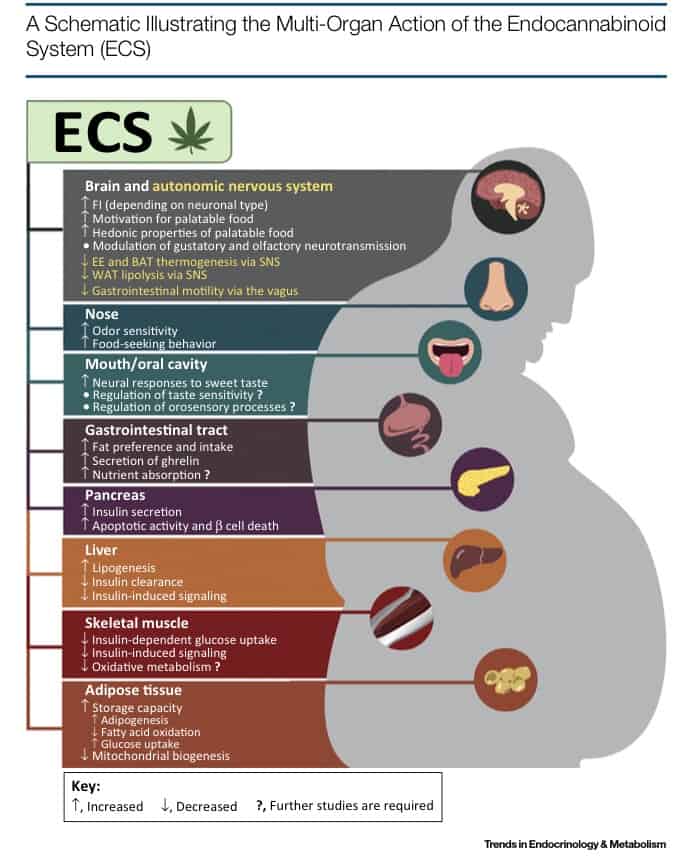
First, the endocannabinoid system is a modulator of the basic senses of smell and taste. Cannabis users may recall the way cannabis can make even the most drab fast food seem exceptionally tasty. In the human body, 2-AG, which is one of the body’s own naturally produced endocannabinoids, “controls odor sensitivity through a CB1 [receptor] dependent mechanism”. Hunger actually stimulates the body to make more 2-AG, which then activates CB1 receptors to make food seem more attractive. Why would this occur? Scientists speculate the system played a role in survival during times when food was scarce. In abundance of food, humans could afford to be picky for the right nutrients and avoidance of potential disease. However, in times of starvation, humans needed to be willing to consume any source of energy possible, and thus hunger, via 2-AG, may have adjusted that desire in response to environmental conditions. This idea is supported by the fact that “malfunctioning of the olfactory system [or sense of smell] has been found in obesity in different organisms”.
However, more than just willingness to eat, the endocannabinoid system can affect fat intake. Studies have shown that endocannabionids produced in the gut can increase or decrease the preferred percentage of fat intake. In fact, blockage of CB1 receptors in the small intestine inhibits fat intake in rodents consistently. Scientists speculate that the nutritional quality and fat quality of food is responsible for changing levels of endocannabinoids in the gut. Why this is occurring remains unclear and needs to be further studied.
Lastly, the endocannabinoid system may control the way fuel is partitioned. In other words, some of the calories that the body intakes are converted into immediately available energy, while others are earmarked for storage, in case energy is needed at a later time when food is scarce. This is actually the holy grail of weight-loss drugs: how can we manipulate the body to give us more energy and less fat? Researchers have discovered that sensory neurons spread throughout fat-storage areas of the body regularly communicate with the brain, which in response, increases or decreases the neurotransmitters that affect both energy levels and appetite. This balance can control our metabolism, and since endocannabinoids can be produced and used quickly by the body, the balance can also be adjusted quickly.
Below we’ve attached a graph illustrating the myriad of effects of the endocannabinoid system in various parts of the body:
So why are we relaying this review to our readers? As we hinted in the second paragraph, the story of endocannabinoids as a weight loss drug is not yet over. The central problem with Rimonabant was not that it didn’t work, but that it had the dangerous side effect of affecting other essential psychiatric processes. Imagine if the endocannabinoids in the gut could be reduced, and that direct stimulation of the brain could be avoided. This is actually possible for CB1 receptor agonists that have a physical structure that prohibits them from crossing the blood-brain boundary. Theoretically, these substances could therefore do what Rimonabant could not and stimulate weight-loss without any negative mental health side effects. Additionally, allosteric modulators, which we reviewed previously and are essentially non-standard activators of CB1 receptors, have also shown promise in reducing caloric intake “without any obvious adverse side effects”. Finally, supplementation of the diet with various enzyme inhibitors can also lead to a downstream decrease in the body’s own endocannabinoids, which can reduce cholesterol and decrease waist-to-hip ratios. In other words, merely deactivating the CB1 receptor all throughout the body is not the only pathway to weight loss; many other roads have yet to be traveled.
Of course, nothing is on the market at the time-being, and if we learned anything from Rimonabant, we can’t be sure at this point in time what other side effects may exist. Smoking cannabis is not an effective weight loss method. However, the potential is definitely there for a truly successful weight-loss drug. Additionally, and perhaps more importantly, diseases that are a result of an overactive endocannabinoid system might also be cured and controlled via substances like these. Only time will tell.
Works Cited
Wilfrid Mazier, Nicolas Saucisse, Blandine Gatta-Cherifi, et al. (2015) The Endocannabinoid System: Pivotal Orchestrator of Obesity and Metabolic Disease. Trends in Endocrinology & Metabolism (2015) 26:524-537.