In general, research into the body’s endocannabinoid system proceeds via administering a substance like a cannabinoid or another drug, and recording the impact of that administration on a specific behavior or physiological process. However, few experiments consider the reverse question: how do our behaviors and environments affect the endocannabinoid system? This system, like most other neurological systems, is not made up of a rigidly fixed number of receptors or ligands. Instead, it constantly changes and adapts to best serve the body, ramping up and down receptor density and receptor affinity as the body deems appropriate.
Stress is now understood to be one of the factors that can impact the endocannabinoid system. In general, exposure to stressful events can easily cause a diverse and lasting set of consequences in both humans and animals. In what is most likely the body’s attempt to overcome and live with chronic stress or the possibility of another stressful event, synapses, which are the paths of communication between individual neurons, re-wire themselves. One extreme example is the maladaptive change that can occur in soldiers put in life or death situations. Soldiers may, for instance, learn to associate sounds heard during such a stressful event with the occurrence of the event itself. Later, in non-hostile environments, these same sounds can then trigger the body and brain to re-live the event, with intense physiological changes, such as the release of adrenaline and increased aggression. This is a classic example of PTSD (Post Traumatic Stress Disorder). However, stress can also change the brain in much more subtle ways, for instance, causing stem cells to malfunction. This can mean shrinkage of various segments of the brain, or decreased ability. Rather than draw problematic connections where there should be none, stress can also cause the brain to eliminate useful, healthy connections and reduce receptor density of various neurological systems. This year, following this line of research, the American College of Neuropsychopharmacology published a review of the various ways stress interacts with the endocannabinoid system.
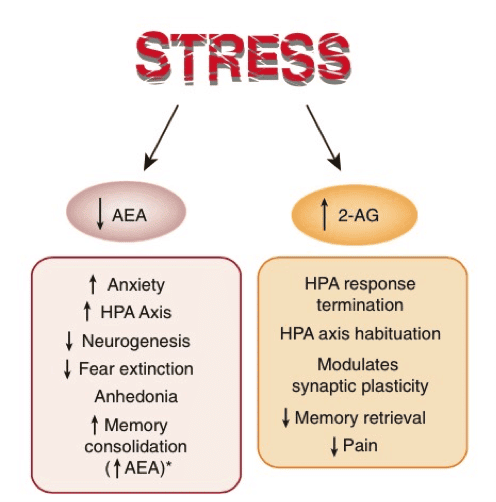
One of the most prominent effects has been shown in the levels of free endocannabinoids circulating in our brain tissues. Readers may recall previous articles on AEA (anandamide) and 2AG (2-arachidonoyl glycerol). We compared AEA, whose name is drawn from the Sanskrit word for “joy”, to THC, since it produces many of the same effects on a shorter duration. However, unlike THC, AEA is produced naturally by the human body and is an essential component of both mental health and the regulation of other important neurological processes. Unfortunately, exposure to stressful events has been shown in multiple experiments to cause circulating levels of AEA to fall. Specifically, within the amygdala, the part of the brain that regulates stress and emotional volatility, stress causes an increase in the level of FAAH, the enzyme that deconstructs and destroys AEA. Likewise, in the hippocampus, which influences memory and the nervous system, acute stress causes a drastic drop in AEA levels as well. In other words, stress can prevent the body from using its own cannabinoids effectively. Think that’s bad? Here’s where it gets really scary: those changes can be permanent. In mice, maternal separation at an early age has been shown to reduce AEA content over the entire lifespan of the rodent! In other areas of the brain, the response is more complex – some brain structures appear to be able to adapt and re-adjust correctly, while others have no response at all. Additionally, strange loopholes exist in regards to stress and AEA levels – foot shock stress on rodents has consistently been shown to increase AEA levels, which is an anomaly that has yet to be explained fully. However, the overall, confirmed trend is that most types of stress decrease AEA levels in multiple areas of the body.
In contrast, stress seems to have a positive effect on 2-AG levels. 2-AG can more accurately be described as “the body’s cannabidiol”, the molecule that primarily activates CB2 receptors. Especially in regards to chronic stress, the brain tends to ramp up circulating 2-AG levels as a means of coping. This effect is more delayed than the reaction of AEA and does not seem to exist in the amygdala, but is consistent in other areas of the brain, like the hippocampus. Likewise, this is thought to be a result of reduction of the enzyme that normally destroys extra 2-AG.
However, aside from affecting levels of circulating endocannabinoids, stress can also cause down-regulation of CB1 receptor density. In multiple areas of the brain, experiments have verified that CB1 receptors disappear under the influence of constant stress. The good news is that unlike the circulating levels of endocannabinoids, this effect is not necessarily permanent and can be reversed following a period of rest and recovery. The areas of the brain in which CB1 receptors are reduced also depend on the duration and type of stress.
Why are these effects occurring? We’re not sure. In regards to AEA and CB1 receptors, the decrease in AEA and CB1 signalling increases anxiety and the acquisition of new memories. On the opposite hand, in regards to 2-AG, studies suggest that the molecule “dampens neural circuits activated by stress” during repeated stress. In both cases, good or bad, endocannabinoid signaling is obviously critical for stress management. But why wouldn’t the body therefore elevate levels of AEA and 2-AG all the time? Why can’t we all just be relaxed and chill 24/7? For better or worse, stress is a motivator of behavior change and activity. Annoyance and frustration, although unpleasant to experience, often yield increased energy over short periods of time. A mouse not stressed by lack of food will subsequently have nothing to eat and starve to death. In other words, the most evolutionarily adaptive response is not necessarily “never being stressed”. The body works to balance responses as appropriate, and the endocannabinoid system is part of that balancing system.
Works Cited
Maria Morena, Sachin Patel, Jaideep Bains, et al. (2015) Neurobiological Interactions Between Stress and the Endocannabinoid System. Neuropsychopharmacology Reviews (2015), 1-23.